Sharing data from clinical trials: the rationale for a controlled access approach – topic of research paper in Clinical medicine. Download scholarly article PDF and read for free on CyberLeninka open science

Overview and experience of the YODA Project with clinical trial data sharing after 5 years | Scientific Data

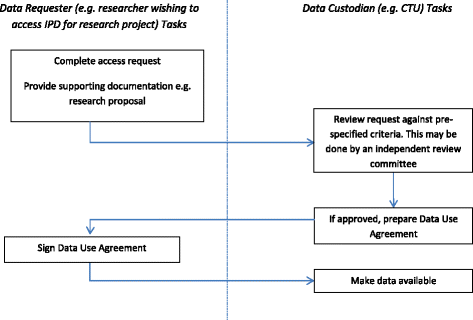
Individual Patient-Level Data Sharing for Continuous Learning: A Strategy for Trial Data Sharing - National Academy of Medicine

Individual Patient-Level Data Sharing for Continuous Learning: A Strategy for Trial Data Sharing - National Academy of Medicine

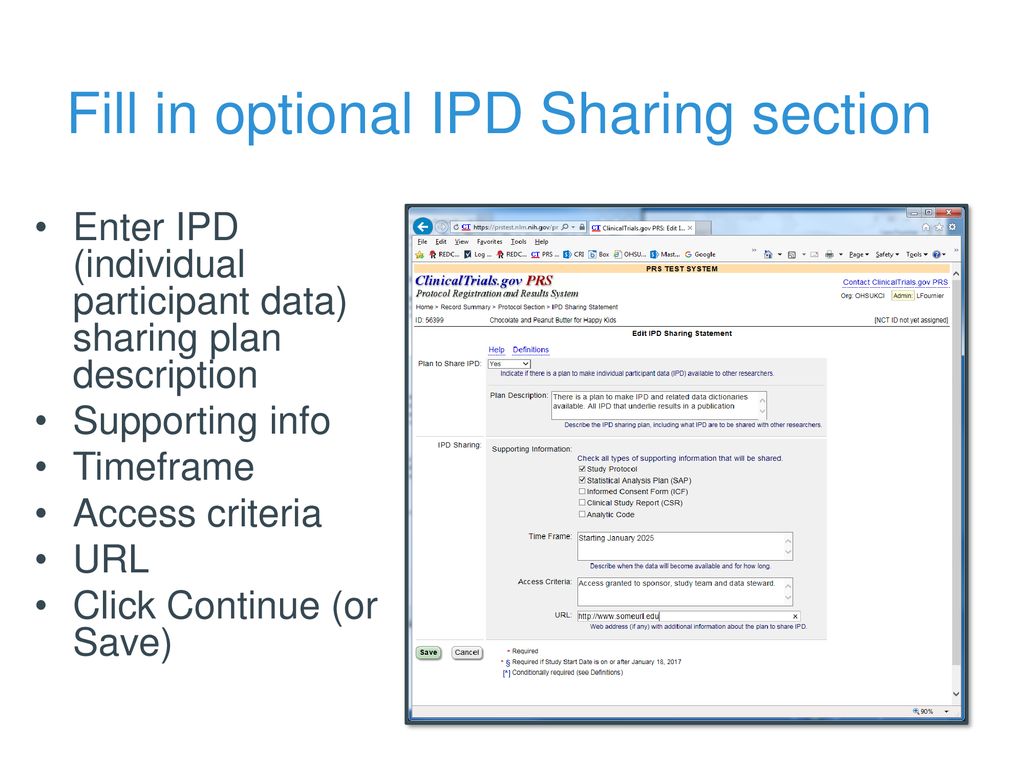
PDSBiggums on Twitter: "$PDSB Interesting new addition to the triple combo study added today. An Individual Participant Data (IPD) Sharing Statement is required in order to have clinical trial results published in

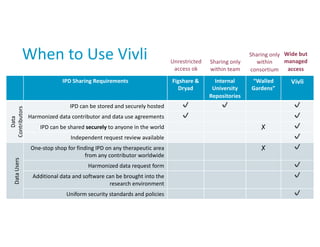
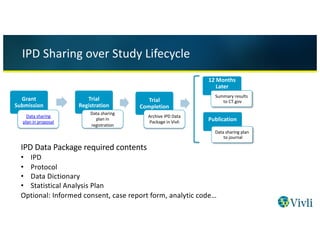
Sharing and reuse of individual participant data from clinical trials: principles and recommendations | BMJ Open